The advantages to using fluorescence vs radioactivity in receptors kinetic and binding assays.

Transform your idea in to molecules
26 January 2023
Game Changer in Target-Based Drug Discovery
7 March 2023
WHY IS IMPORTANT TO CONSIDER MULTIPLE ASSAY TYPES FOR TARGETS CHARACTERIZATION?
Usually, in the identification of new drug-like molecules for a target of interest it is necessary to determine its affinity for the target. Radioactive ligands are commonly used to measure ligand binding to receptors both in whole cells and cell membranes. Radioligands can be used to perform saturation curves, competition and kinetic experiments providing information about receptor expression (Bmax), binding affinity of ligand (KD , Ki ), IC50 , Kon and Koff .
The allosteric nature of GPCRs dictates a bidirectional flow of allosteric energy between the extracellular ligand binding site and the binding sites in the cytoplasm for signalling molecules. When a single probe of the receptor is used to detect ligand interaction with that receptor (i.e., radioligand), then any ligands that interact with the receptor but do not report that interaction to the screening probe, will be missed (i.e., many allosteric ligands). In contrast, a functional assay utilizes the full frame of signalling proteins in the cell interrogating the receptor and reporting alteration of receptor conformation and function when ligands bind.
Fluorescent ligands represent an effective and more versatile alternative to help researchers fully characterize specific GPCRs, combining affinity and functional screening assays.
RADIOLIGANDS
Radioligand assays are useful due to the high chemical similarity of unlabeled and labelled probe molecules and the highly specific detection of radioactive label. However, radioligand-based methods have several disadvantages such as the expensive costs, generation of large quantities of radioactive waste and problems associated with the disposal of it and the regulatory requirements associated.
Furthermore, high-affinity radioligands are not available for all the targets of interest and their stability in some cases can represent an issue. For these reasons over the last years, a great effort was put into the generation of cost-effective, risk-averse and environmentally friendly alternatives to radioligand assays.
FLUORESCENT LIGANDS
Fluorescence ligands are useful tools for kinetic and binding assays independently of antagonist/agonist activity and can be used for the determination of target expression levels they can provide another advantage in several fluorescence techniques (fluorescence polarization, scanning confocal microscopy, fluorescence spectroscopy, fluorescence resonance energy transfer and flow cytometry).
These fluorescent techniques provide a better understanding of receptor localization at the cellular and subcellular levels, function and regulation. They allow the real-time visualization of the labelled receptor and the detection of internalization, endocytosis and recycling process. They can be used as a tool to study oligomerization processes and they offer the possibility to detect colocalization by using multilabelling strategies. In general, they can be adapted to run in high-throughput formats.
THE BRIGHT FUTURE OF FLUORESCENT LIGANDS
High-affinity fluorescent antagonists can be used to label the target receptors, and fluorescently tagged agonists can provide a means to monitor dynamic processes such as receptor internalization and trafficking (1). A fluorescent ligand in addition to being used as the competing probe in a competition-based binding assay can be implemented to measure the direct displacement of a competing fluorescent ligand from a GPCR orthosteric site. These alternatives demonstrated enormous potential for revealing elaborate and intricate details about receptor oligomerization through the use of FRET and BRET assays (4).
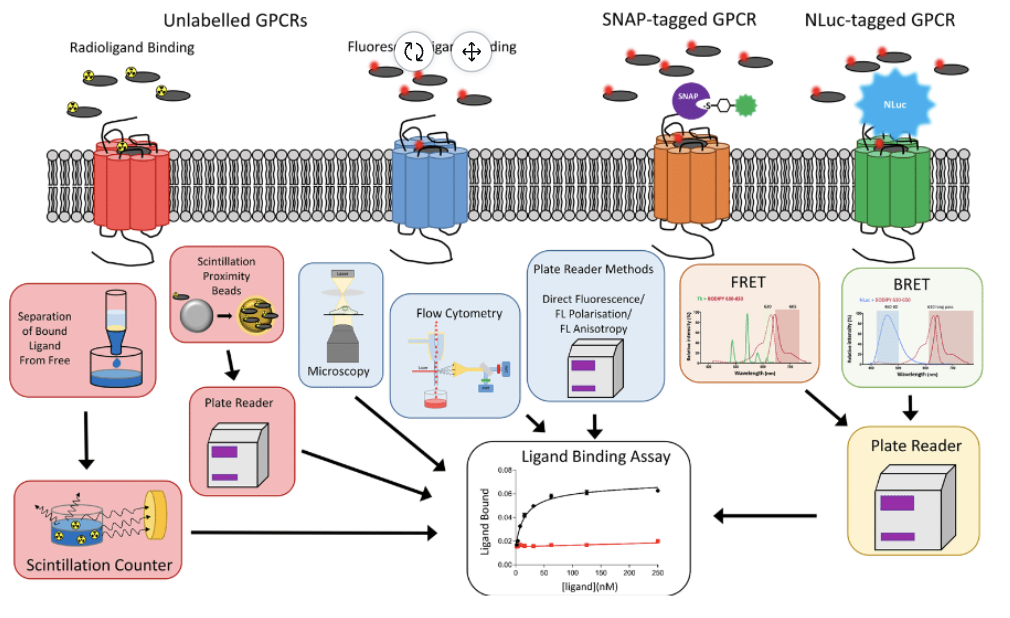
WHAT YOU CAN DO WITH RADIOLIGANDS
Using untagged receptors, radioligand-binding assays rely on the use of scintillation proximity technologies, or the separation of the bound ligand from free ligand, followed by the detection of bound ligand using scintillation counters.
WHAT YOU CAN DO WITH
FLUORESCENT LIGANDS
Binding to unlabelled GPCR can be monitored through microscopy, using plate readers to detect fluorescence, or through fluorescent polarization and flow cytometry. Alternatively, the binding of fluorescent ligands to SNAP-tagged or NanoLuc-tagged GPCRs can be detected with RET approaches (FRET and BRET, respectively).
WHY USE THE FLUORESCENT LIGANDS OF CELTARYS
Fluorescence dyes can also have disadvantages as usually fluorescent labels are larger than radioisotopes thus altering the activity of ligands.
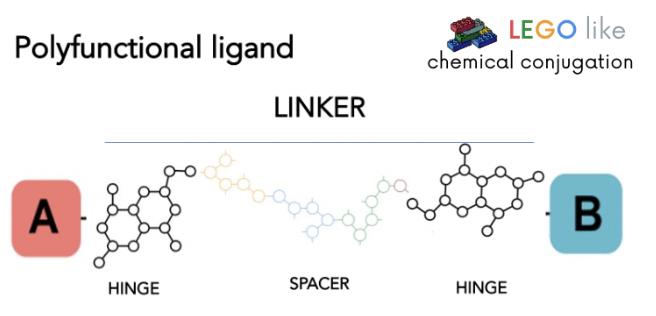
The main advantage that can offer Celtarys LEGO-like chemical conjugation technology with regards to other fluorescent ligands is the chance to develop tailor-made fluorescent ligands to use in the optimization of a wide set of technologies for studying the majority of therapeutic targets.
We offer more than 400 different linkers architectures to combine with a large set of fluorophores, opening the opportunity to generate a higher chemical space diversity and a higher chance to identify candidates with good physiochemical and bioactivity properties.
REFERENCES
- Soave, M., Briddon, S. J., Hill, S. J., & Stoddart, L. A. (2020). Fluorescent ligands: Bringing light to emerging GPCR paradigms. British Journal of Pharmacology, 177(5), 978-991.
- Barbazán, J., Majellaro, M., Martínez, A. L., Brea, J. M., Sotelo, E., & Abal, M. (2022). Identification of A2BAR as a potential target in colorectal cancer using novel fluorescent GPCR ligands. Biomedicine & Pharmacotherapy, 153, 113408.
- Albizu, L., Holloway, T., González-Maeso, J., & Sealfon, S. C. (2011). Functional crosstalk and heteromerization of serotonin 5-HT2A and dopamine D2 receptors. Neuropharmacology, 61(4), 770-777.
- Hill, S. J., May, L. T., Kellam, B., & Woolard, J. (2014). Allosteric interactions at adenosine A1 and A3 receptors: new insights into the role of small molecules and receptor dimerization. British Journal of Pharmacology, 171(5), 1102-1113.
- May, L. T., Bridge, L. J., Stoddart, L. A., Briddon, S. J., & Hill, S. J. (2011). Allosteric interactions across native adenosine-A3 receptor homodimers: quantification using single-cell ligand-binding kinetics. The FASEB Journal, 25(10), 3465.
- Briddon, S. J., & Hill, S. J. (2007). Pharmacology under the microscope: the use of fluorescence correlation spectroscopy to determine the properties of ligand–receptor complexes. Trends in pharmacological sciences, 28(12), 637-645.





