Fluorescent ligands: A New Method to Label your GPCRs

Celtarys Adenosine Receptor fluorescent ligands as potent tools for the study of cancer specific GPCRs
26 July 2022
Video webinar – Celtarys Colorectal Cancer & GPCR Fluorescent ligands
26 October 2022To date, it has been reported that about 800 GPCRs are encoded by the human genome. Some of them have known ligands available which led to the corresponding radioligands. However, in most cases they are orphan receptors, which means that no ligands are known, or there are only primary antibodies available for their study. When directly labelled or using labelled secondary antibodies (indirect immunofluorescence), primary Abs can be used to detect the GPCRs expression through a wide range of techniques, including immunohistochemistry, proximity ligation assays and immunoblotting.
Unfortunately, the generation of antibodies against unmodified GPCRs (as endogenously expressed GPCRs) is not a simple task because there are several significant hurdles to overcome:
- Low cell surface expression.
- Need for the GPCR to be expressed in a membrane with the correct post-translational modifications.
- Intrinsic conformational heterogeneity of GPCRs.
- Low immunogenicity, with only the N-terminal domain and extracellular loops accessible as potential epitopes for extracellularly targeted antibodies.
Generating high specific antibodies can be a complex task and, especially for GPCRs, it can be more challenging.
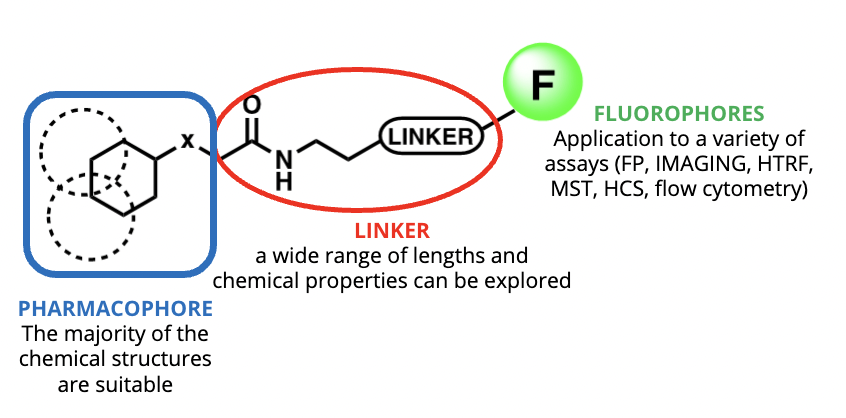
It is important to remember that one of the inherent properties of a GPCR is to bind small molecules or peptides. Using these innate physiological properties, fluorescently labelled GPCR ligands represent an alternative to antibodies to detect endogenously expressed receptors. These fluorescent tools are composed by a pharmacophore which binds the receptor of interest, a linker and a fluorophore.
In the following paper, we describe the biggest advantages of using fluorescent ligands for GPCR characterization, combining different techniques in the same experiment.
If you want to know more about how to use fluorescent ligands to characterize receptors with pharmacological interest with this and other technologies, click on the button to download the article “Fluorescent ligands: A new method to label your GPCRs”
Download the full article




